|
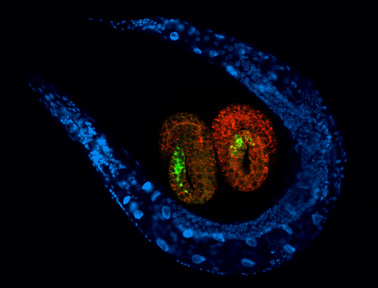
C. elegans - nature's gift to science - Sydney Brenner, 2002 Nobel Prize seminar Being the first organism whose genome sequence was entirely known, C. elegans has been one of the leading genetic models for biological research. C. elegans is the only organism whose entire developmental lineage was elucidated at single-cell resolution, the connectivity of the nervous system was deciphered by serial EM section plus reconstruction, and the first organism to glow engineered GFP in live. The vision, insights and collegiality of the C. elegans community worldwide make working with this elegant nematode an enduring and inspiring pleasure. |
Our lab is broadly interested in developmental neuroscience and the biological basis of nervous system aging. We use the soil-living nematode Caenorhabditis elegans as a model organism, taking advantage of its powerful genetic tools. Moreover, neuronal circuitry and synaptic connections of the C. elegans nervous system had been mapped to single-cell resolution, which greatly facilitates studies of the function of individual neurons in unique behavioral contexts. We are particularly interested in learning, memory, behavioral control and neural mechanisms for stress and physiological regulation.
Current Projects
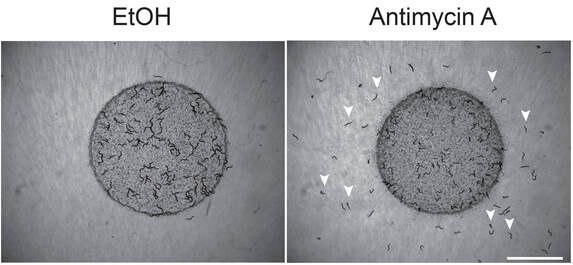
Aversive Learning under Stress
Animal behavior can be modified by past experience and internal states, which constitute the basis of learning and memory. Internal states such as feeding status, sex drive and hormonal profiles are powerful influences that profoundly change animal behavior. Physiological stress, including mitochondrial disruption, defective proteostasis or inflammation, also induces aversive learning and avoidance behavior. Stress-induced aversive learning is particularly important for the animals to avoid potential threat and dangers in order to optimize their chance of survival in the wild. Using mitochondrial disruption as a stressor, we found that mitochondrial stress triggered bacterial avoidance in C. elegans, and this avoidance showed featured of long-term associative memory of higher organisms. We further uncovered several neuronal types and secreted signals that govern this aversive associative memory and behavior (Neurosci Res 2022, 2023; PNAS 2022; Current Biology 2022). Using genetic, molecular, imaging and behavioral approaches combined with high throughput gene expression profiling, we aim at elucidating the circuit and molecular mechanisms of stress-induced memory and learning.
Animal behavior can be modified by past experience and internal states, which constitute the basis of learning and memory. Internal states such as feeding status, sex drive and hormonal profiles are powerful influences that profoundly change animal behavior. Physiological stress, including mitochondrial disruption, defective proteostasis or inflammation, also induces aversive learning and avoidance behavior. Stress-induced aversive learning is particularly important for the animals to avoid potential threat and dangers in order to optimize their chance of survival in the wild. Using mitochondrial disruption as a stressor, we found that mitochondrial stress triggered bacterial avoidance in C. elegans, and this avoidance showed featured of long-term associative memory of higher organisms. We further uncovered several neuronal types and secreted signals that govern this aversive associative memory and behavior (Neurosci Res 2022, 2023; PNAS 2022; Current Biology 2022). Using genetic, molecular, imaging and behavioral approaches combined with high throughput gene expression profiling, we aim at elucidating the circuit and molecular mechanisms of stress-induced memory and learning.
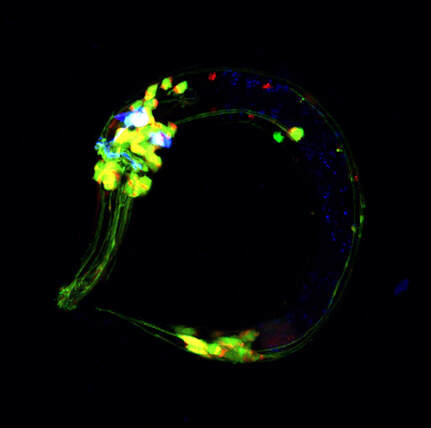
Gut-Brain Signaling in Behavioral Plasticity
Animal behavior is influenced by internal states. Internally-derived signals encode the physiological states of nutrition, metabolism and tissue integrity, and they act upon the nervous system to induce behavioral plasticity. So-called "gut-brain signaling", this communication from somatic tissues to the brain has wide implications for physiology, behaviors and diseases. We are currently using genetic, genomic and biochemical approaches (in collaboration with labs in the US and Europe) to identify these "state-encoding" signals to understand the mechanistic details and biological significance of gut-brain signaling for stress-induced aversive learning. Our results show that metabolic remodeling under physiological stress is a critical step in generating a wide variety of tissue signals for behavioral plasticity and learning. This exciting aim of research will open the door to understanding how the nervous system and the somatic tissues communicate in a bi-directional manner for both physiological fitness, learning and behavior.
Animal behavior is influenced by internal states. Internally-derived signals encode the physiological states of nutrition, metabolism and tissue integrity, and they act upon the nervous system to induce behavioral plasticity. So-called "gut-brain signaling", this communication from somatic tissues to the brain has wide implications for physiology, behaviors and diseases. We are currently using genetic, genomic and biochemical approaches (in collaboration with labs in the US and Europe) to identify these "state-encoding" signals to understand the mechanistic details and biological significance of gut-brain signaling for stress-induced aversive learning. Our results show that metabolic remodeling under physiological stress is a critical step in generating a wide variety of tissue signals for behavioral plasticity and learning. This exciting aim of research will open the door to understanding how the nervous system and the somatic tissues communicate in a bi-directional manner for both physiological fitness, learning and behavior.
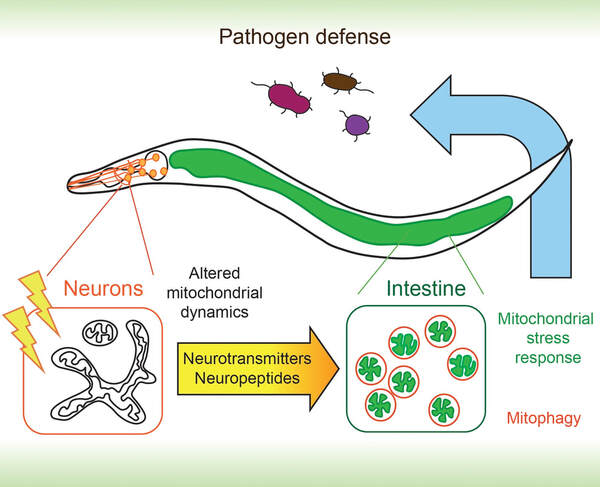
Neural Control of Systemic Mitochondrial States
We study how neuronal signals regulate mitochondrial states at the organismal level. The significance of this control could be seen in stress response, pathogen resistance and aging. Our recent study has identified a network of shared and distinct neurotransmitters and neuropeptides that regulate mitochondrial morphology and stress responses in distal tissues, such as the intestine (Dev Cell 2021). mRNA sequencing experiments further uncovered distinct gene sets that contribute to enhanced immunity under neuronal signals. We are currently elucidating the molecular mechanisms that transduce neuronal signals to regulate mitochondrial states.
We study how neuronal signals regulate mitochondrial states at the organismal level. The significance of this control could be seen in stress response, pathogen resistance and aging. Our recent study has identified a network of shared and distinct neurotransmitters and neuropeptides that regulate mitochondrial morphology and stress responses in distal tissues, such as the intestine (Dev Cell 2021). mRNA sequencing experiments further uncovered distinct gene sets that contribute to enhanced immunity under neuronal signals. We are currently elucidating the molecular mechanisms that transduce neuronal signals to regulate mitochondrial states.
Past Projects
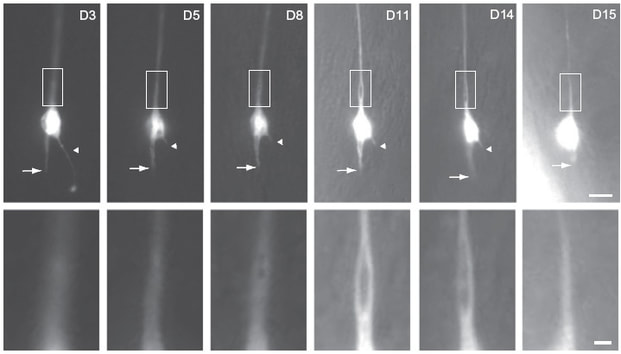
Cell biology of neuronal aging.
The molecular mechanisms leading to the dismantling of apoptotic cells and injured neurons during development had been studied in detail. However, our understanding of the cell biological events occurring in the process of neuronal aging is far from clear. Work from our lab suggest that C. elegans neurons and mitochondria undergo characteristic morphological changes in senescence (PNAS 2011, PNAS 2015). Recently we collaborated with the lab of Ikue Mori to investigate the functional property of neurons during aging (Aging Cell 2020). We are now using various genetic and cell biological approaches to investigate how these events are regulated at the molecular and organelle level. (Photo courtesy of PNAS)
The molecular mechanisms leading to the dismantling of apoptotic cells and injured neurons during development had been studied in detail. However, our understanding of the cell biological events occurring in the process of neuronal aging is far from clear. Work from our lab suggest that C. elegans neurons and mitochondria undergo characteristic morphological changes in senescence (PNAS 2011, PNAS 2015). Recently we collaborated with the lab of Ikue Mori to investigate the functional property of neurons during aging (Aging Cell 2020). We are now using various genetic and cell biological approaches to investigate how these events are regulated at the molecular and organelle level. (Photo courtesy of PNAS)
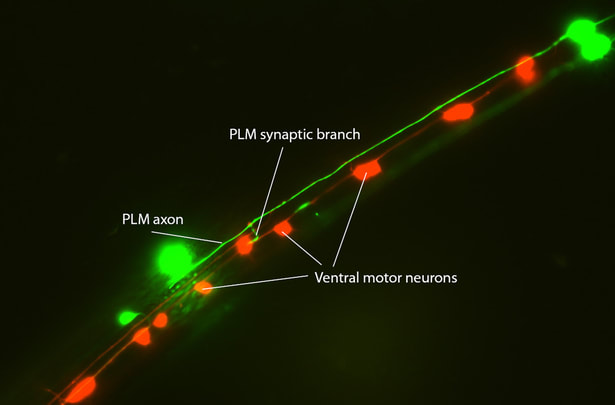
Molecular mechanisms of axon and dendrite development.
Axons and dendrites often elaborate branches during development. These branches shape the connectivity of neuronal circuits and lay the foundation for behaviors. We identify RHGF-1, a microtubules-associated RhoGEF, as a mediator of branch retraction following microtubule disassembly (PNAS 2014). By characterizing a gain-of-function tubulin mutant, we also demonstrate the electrostatic coupling between microtubules and dynein motor, which is an important step in neuronal polarization (PLoS Genet 2014). We further show that Wnt signaling instructs neurite branching by remodeling F-actin dynamics (PLoS Genet 2017), and it can be tuned by a polarity protein, VANG-1, for tightly controlling neuronal migration (Development 2018). VANG-1 also controls dendrite development through modulating Wnt signaling activity (Development 2020). Surprisingly, the Wnt-secretory factor MIG-14/Wntless regulates dendrite self-avoidance, but this is independent of Wnt functions, revealing an unexpected role for Wntless (Neuron 2018). Dendrites can develop and extend by attaching to a scaffold axon, and this axon-dendrite fasciculation is critical for the structure and function of the somatosensory neuronal circuit (Dev Cell 2019).
Axons and dendrites often elaborate branches during development. These branches shape the connectivity of neuronal circuits and lay the foundation for behaviors. We identify RHGF-1, a microtubules-associated RhoGEF, as a mediator of branch retraction following microtubule disassembly (PNAS 2014). By characterizing a gain-of-function tubulin mutant, we also demonstrate the electrostatic coupling between microtubules and dynein motor, which is an important step in neuronal polarization (PLoS Genet 2014). We further show that Wnt signaling instructs neurite branching by remodeling F-actin dynamics (PLoS Genet 2017), and it can be tuned by a polarity protein, VANG-1, for tightly controlling neuronal migration (Development 2018). VANG-1 also controls dendrite development through modulating Wnt signaling activity (Development 2020). Surprisingly, the Wnt-secretory factor MIG-14/Wntless regulates dendrite self-avoidance, but this is independent of Wnt functions, revealing an unexpected role for Wntless (Neuron 2018). Dendrites can develop and extend by attaching to a scaffold axon, and this axon-dendrite fasciculation is critical for the structure and function of the somatosensory neuronal circuit (Dev Cell 2019).
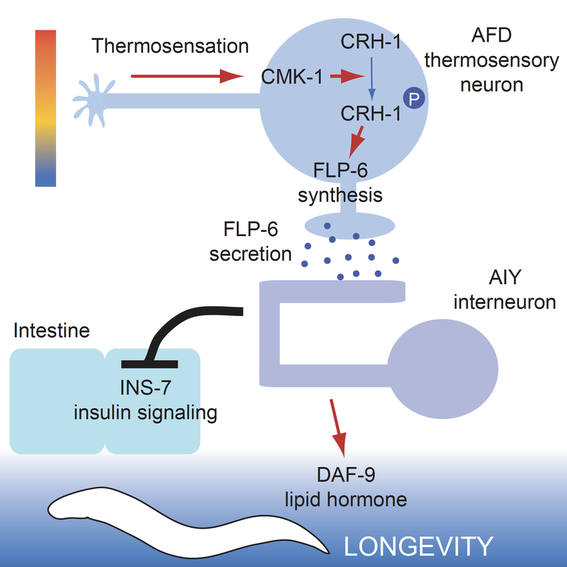
Regulation of systemic physiology and longevity by the nervous system.
The nervous system in C. elegans, as that in more complex organisms, plays a critical role in maintaining systemic homeostasis of physiology. Our recent work shows that the thermosensory neuronal circuit controls C. elegans longevity through a secreted neuropeptide (Dev Cell 2016). C. elegans has a conserved panel of neurotransmitters (ACh, GABA, glutamate, dopamine, 5HT, octopamine, tyramine) and an unusually rich repertoire of neuropeptides, with only a handful being functionally characterized. We are in the process of deciphering the molecular and circuit mechanisms through which neurons exert cell non-autonomous regulation of physiology and aging in distal tissues.
The nervous system in C. elegans, as that in more complex organisms, plays a critical role in maintaining systemic homeostasis of physiology. Our recent work shows that the thermosensory neuronal circuit controls C. elegans longevity through a secreted neuropeptide (Dev Cell 2016). C. elegans has a conserved panel of neurotransmitters (ACh, GABA, glutamate, dopamine, 5HT, octopamine, tyramine) and an unusually rich repertoire of neuropeptides, with only a handful being functionally characterized. We are in the process of deciphering the molecular and circuit mechanisms through which neurons exert cell non-autonomous regulation of physiology and aging in distal tissues.